DIFFUSION
- the movement of individual molecules of a substance through a semipermeable barrier from an area of higher concentration to an area of lower concentration
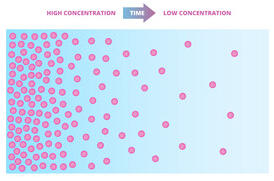

Small molecules move across a cell membrane through a process called diffusion. Diffusion usually takes place in liquids like water and gases like air where the molecules moves from high concentration to low.
⋆ air particles possess energy
⋆ particles start moving randomly
⋆ they bump and bounce into one another
⋆ overtime, air particles spread out and fill the entire container
what affects the rate of diffusion?
Diffusion happens faster in gases because the particles are more spread out and allows the molecules to move faster.Temperature can also affect the rate of diffusion. The hotter the temperature, the faster the particles spread because there's more energy.The larger the volume of space and lesser the surface area, the lower the rate of diffusion. There is a greater area that needs to receive the substance being diffused, but less area for that substance to actually enter.
basic examples of diffusion:
◇ a common example is gas exchange in the lungs, where the oxygen is moving into blood cells and carbon dioxide is moving into the alveoli◇ food colouring diffusing into water
◇ water diffusing into vinegar-soaked egg
◇ sugar diffusing into coffee
◇ water diffuses into plants
◇ coffee smell diffusing throughout the coffee shopA sugar cube begins to dissolve when dropped into water; the molecules move from a high concentration into a low concentration area and is distributed evenly, effectively diffusing.